Examine The Following Phase Diagram And Determine What Phase Exists At Point D
Sign up to view the full version. Vapor which of the following intermolecular forces is the weakest.
 Phase Changes Boundless Chemistry
Phase Changes Boundless Chemistry
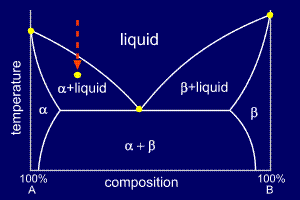
If we change the composition of the liquid or the temperature the number of phases will be reduced to 2.
Examine the following phase diagram and determine what phase exists at point d. C bo changes from a solid to a liquid as one follows the line from c to d. Increasing temperature with a phase change from solid to vapor examine the following phase diagram and determine what phase exists at point f. The eutectic point is therefore an invariant point.
E point b represents the critical temperature and pressure for bo. Answer to examine the following phase diagram and determine what phase exists at point c. Examine the phase diagram for the substance bogusium bo and select the correct statement.
B the triple point for bo is at a higher temperature than the melting point for bo. B the triple point for bo is at a higher temperature than the melting point for bo. Strontium metal crystallizes in a cubic unit cell which has an edge length of 612 pm.
If the the density of sr metal is 254 gcm3 calculate the number of atoms per unit cell. Examine the following phase diagram and identify the. Examine the phase diagram for the substance bogusium bo and select the correct statement.
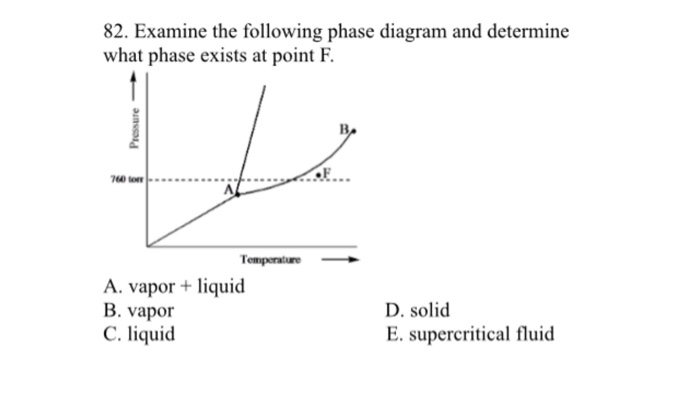
Since we looking at a system at constant pressure the phase rule in this case is f c 1 p. Gas and liquid 2. Examine the following phase diagram and determine what phase exists at point 760 som temperature a supercritical fluid b liquid c vaporliquid d vapor e solid the phase diagram of a substance i s given below.
Chapter 12 consider the following phase diagram and identify the process occurring as one goes from point c to point d. Liquids solids and phase changes 15. C bo changes from a solid to a liquid as one follows the line from c to d.
See question 10 image a bos has a lower density than bol. Examine the following phase diagram and identify the feature represented by point a. Consider the following phase diagram and identify the process occurring as one goes from point c to point d.
This preview has intentionally blurred sections. Consider the following phase diagram and identify the process occurring as one goes from point c to point d. Examine the following phase diagram and identify the feature represented by point a.
What type of unit cell is this. Aincreasing temperature with a phase change from solid to liquid bincreasing temperature with a phase change from solid to vapor cincreasing temperature with a phase change from liquid to vapor dincreasing temperature with no phase change eincreasing temperature beyond the critical point7. A bos has a lower density than bol.
Examine the phase diagram for the substance bogusium bo and select the correct statement.
 Phase Change Evaporation Condensation Freezing Melting
Phase Change Evaporation Condensation Freezing Melting
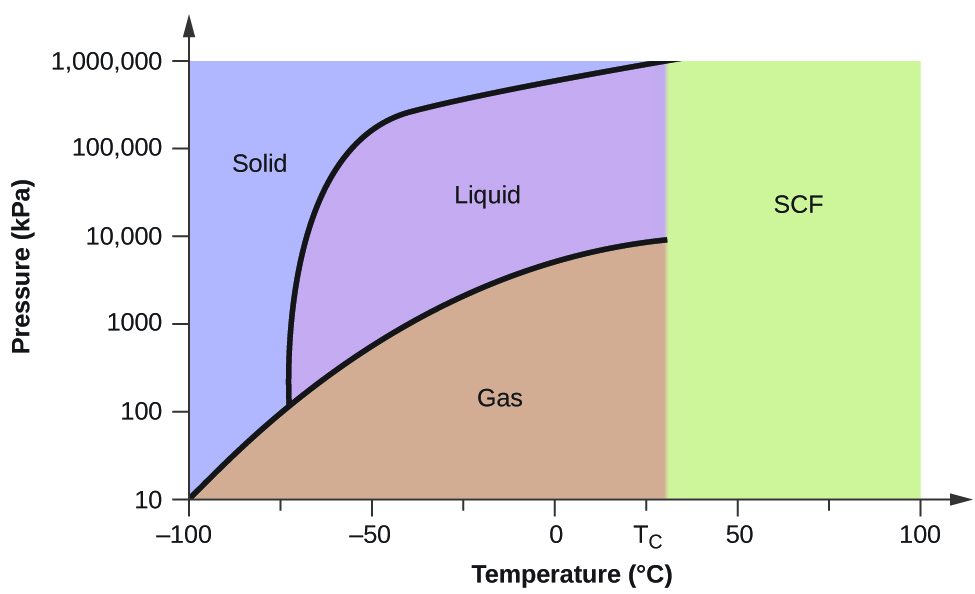
 Phase Diagrams Of Water Co2 Explained Chemistry Melting Boiling Critical Point
Phase Diagrams Of Water Co2 Explained Chemistry Melting Boiling Critical Point
 Phase Diagrams An Overview Sciencedirect Topics
Phase Diagrams An Overview Sciencedirect Topics
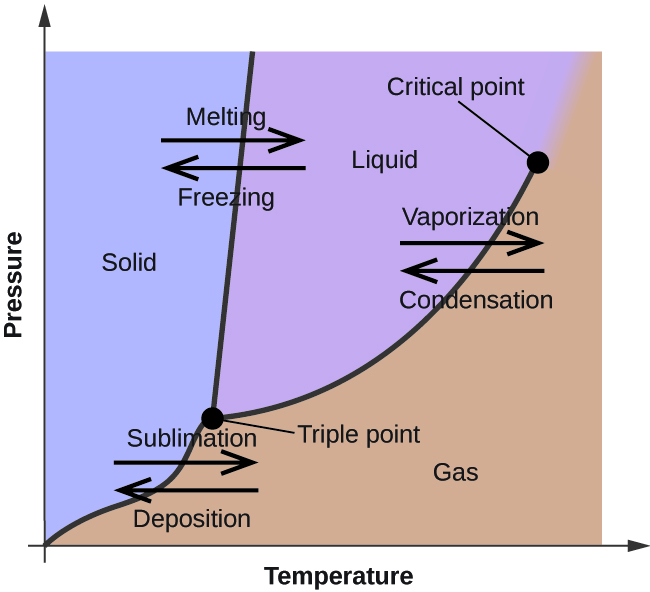
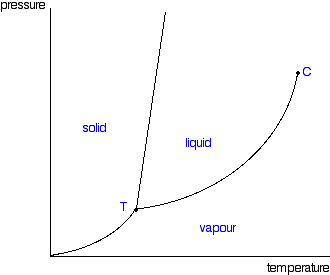 Phase Diagrams Of Pure Substances
Phase Diagrams Of Pure Substances
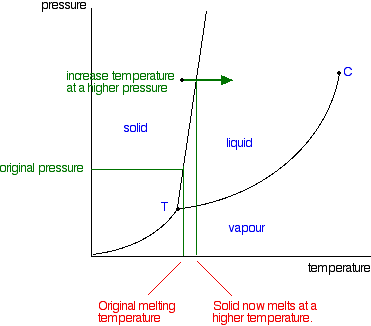 Phase Diagrams Of Pure Substances
Phase Diagrams Of Pure Substances
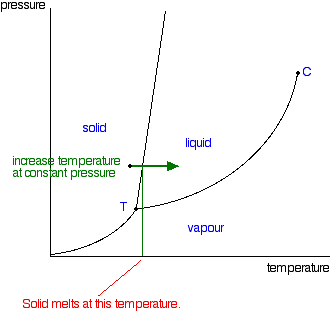 Phase Diagrams Of Pure Substances
Phase Diagrams Of Pure Substances
 Ammonia Vapour Pressure At Gas Liquid Equilibrium
Ammonia Vapour Pressure At Gas Liquid Equilibrium
 Phase Diagrams An Overview Sciencedirect Topics
Phase Diagrams An Overview Sciencedirect Topics
 Selected Answer B 444 G Ar Correct Answer B 444 G Ar
Selected Answer B 444 G Ar Correct Answer B 444 G Ar
 Comparisons Between Phase Diagrams And The Hill And Valley
Comparisons Between Phase Diagrams And The Hill And Valley
 Delta Iron An Overview Sciencedirect Topics
Delta Iron An Overview Sciencedirect Topics
 Thermodynamic Phase Diagram Of Amyloid B 16 22 Peptide Pnas
Thermodynamic Phase Diagram Of Amyloid B 16 22 Peptide Pnas

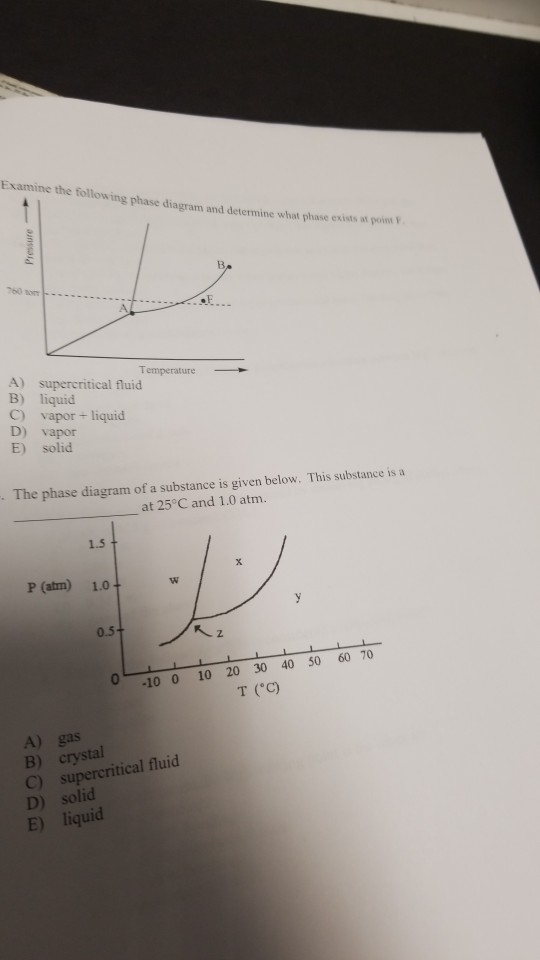 Solved Examine The Following Phase Diagram And Determine
Solved Examine The Following Phase Diagram And Determine
Considerations And Challenges In Studying Liquid Liquid





0 Response to "Examine The Following Phase Diagram And Determine What Phase Exists At Point D"
Post a Comment