H2 Molecular Orbital Diagram
Bonding and anti bonding molecular orbitals in h 2. If the electrons are in phase they have a constructive interference.
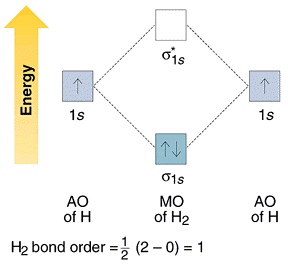
This results in a bonding sigma mo σ 1s.
H2 molecular orbital diagram. This mo has an increased probability of finding electrons in the bonding region. Molecular orbital theory posits the notion that electrons in molecules likewise exist in different orbitals that give the probability of finding the electron at particular points around the molecule. Molecular orbital mo theory of the h2 molecule.
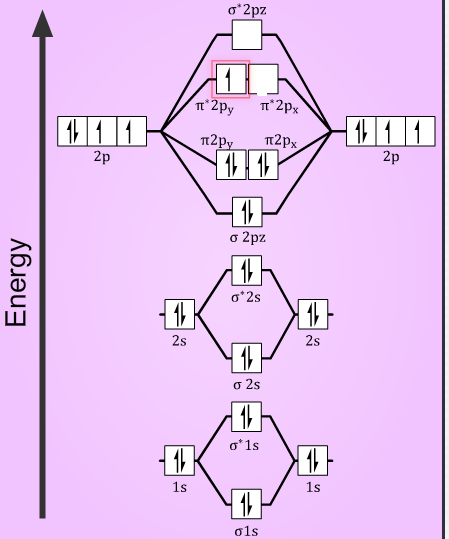
A diatomic molecular orbital diagram is used to understand the bonding of a diatomic molecule. πε and jr k r mr lr defined explicitly in atkins. Fill from the bottom up with 2 electrons total.
They also give insight to the bond order of the molecule how many bonds are shared between the two atoms. To produce the set of orbitals for a molecule we add together the valence atomic wavefunctions for the bonded atoms in the molecule. Ab a b gg with.
Description of the molecular orbitals of the h2 molecule with an introduction to molecular orbital diagrams. Its molecular orbitals are constructed from the valence shell orbitals of each hydrogen atom which are the 1 s orbitals of the atoms. Discussed in this video are.
Some facts are known about hydrogen due to some careful experimental measurements. Bonding mos antibonding mos and bond order. Here is the result obtained for egs r via eq.
Molecular orbital mo theory suggested atomic orbitals which not only overlap but are simultaneously transformed into molecular orbitals with new energies and new electron distributions. Bonding order is 1 and it is diamagnetic. The qualitative approach of mo analysis uses a molecular orbital diagram to visualize bonding interactions in a molecule.
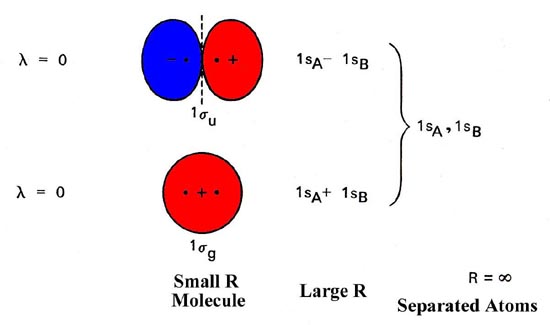
Mo diagrams can be used to deduce magnetic properties of a molecule and how they change with ionization. When two h atoms come to a proper proximity their 1s orbitals interact and produce two molecular orbitals. Molecular orbitals of h 2 and he2.
A bonding mo and an anti bonding mo. Two superpositions of these two orbitals can be formed one by summing the orbitals and the other by taking their difference. In this type of diagram the molecular orbitals are represented by horizontal lines.
Since both molecular ions have a bond order of 12 they are approximately equally stable. Molecular orbital diagram for hydrogen gas h2. Surprisingly the hybridization of the starred oxygen in the following molecule is sp 2 not sp 3.
Evaluate the ground state electronic energy based on this presumed approximate eigenfunction. The higher a line the higher the energy of the orbital and degenerate orbitals are placed on the same level with a space between them. Each h atom has a 1s atomic orbital.
 Molecular Orbitals Molecular Orbitals For Homonuclear
Molecular Orbitals Molecular Orbitals For Homonuclear
 11 5 Molecular Orbital Theory Chemistry Libretexts
11 5 Molecular Orbital Theory Chemistry Libretexts
 Artificial Nodes In The H2 Wave Functions Expanded Using
Artificial Nodes In The H2 Wave Functions Expanded Using
 9 7 Molecular Orbitals Chemistry Libretexts
9 7 Molecular Orbitals Chemistry Libretexts
 Molecular Orbitals Of H2 Chemistry Libretexts
Molecular Orbitals Of H2 Chemistry Libretexts
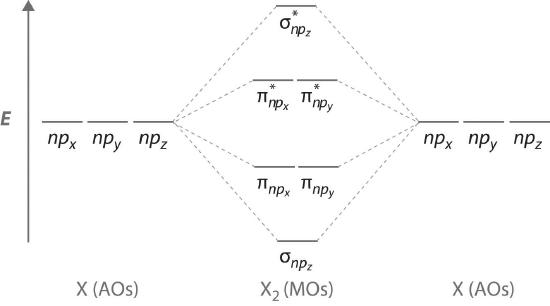 9 7 Molecular Orbitals Chemistry Libretexts
9 7 Molecular Orbitals Chemistry Libretexts
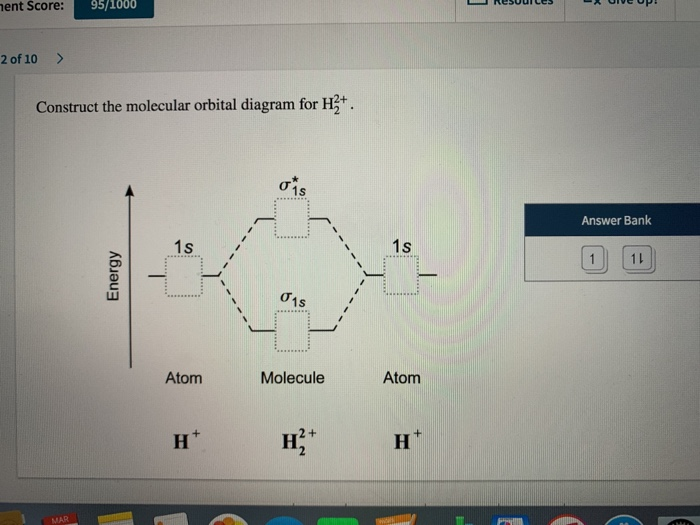 Solved Ent Score 95 1000 2 Of 10 Construct The Molecula
Solved Ent Score 95 1000 2 Of 10 Construct The Molecula
Molecular Orbital Theory Mot Chemistry Study Material
Lecture 3 January 9 2015 Bonding In H2
Bohr S Hydrogen Molecule Ion H2
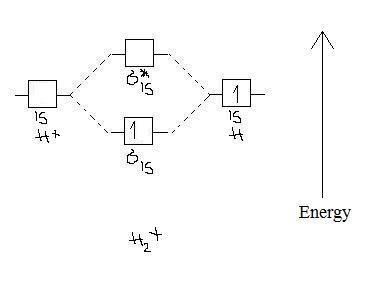 2 3b Mo Theory Of Bonding In H Chemistry Libretexts
2 3b Mo Theory Of Bonding In H Chemistry Libretexts
14 Linear Combination Of Atomic Orbitals Chemistry Libretexts
 100 Molecular Orbital H Yasminroohi
100 Molecular Orbital H Yasminroohi
 Bonding In Some Homonuclear Diatomic Molecules Emedicalprep
Bonding In Some Homonuclear Diatomic Molecules Emedicalprep
Lecture 3 January 9 2015 Bonding In H2
 Solved Consider The H2 Ion A Sketch The Molecular
Solved Consider The H2 Ion A Sketch The Molecular
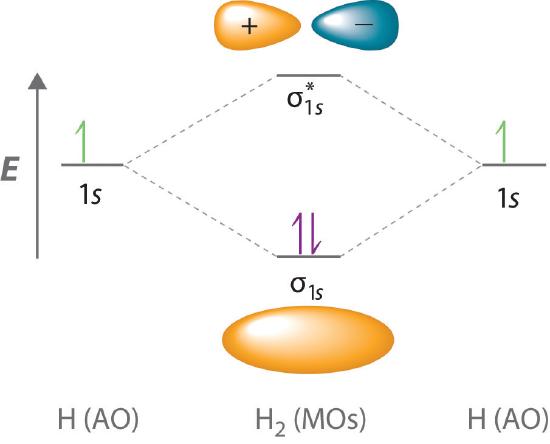 9 7 Molecular Orbitals Chemistry Libretexts
9 7 Molecular Orbitals Chemistry Libretexts
 How Do You Write The Electron Configuration For H H 2
How Do You Write The Electron Configuration For H H 2
 What Is The Molecular Orbital Energy Diagram Of Co Quora
What Is The Molecular Orbital Energy Diagram Of Co Quora
 Pdf Solutions Manual To Accompany Inorganic Chemistry Sixth
Pdf Solutions Manual To Accompany Inorganic Chemistry Sixth
Which One Is More Stable H2 Or H2 Quora
 Energy Level Diagram For Molecular Orbitals Chemical
Energy Level Diagram For Molecular Orbitals Chemical
 H2 Molekularen Oinarrizko Energia
H2 Molekularen Oinarrizko Energia
0 Response to "H2 Molecular Orbital Diagram"
Post a Comment