Molecular Orbital Diagram For He2
Construct the molecular orbital diagram for he2 and then identify the bond order. Ionization potential curves plotted on the diagrams for the neutral systems are nearly parallel to the corresponding orbital energy curves.
 Molecular Orbital Theory Grandinetti Group
Molecular Orbital Theory Grandinetti Group
This explanation is based on a mathematical model hence it is a theory.
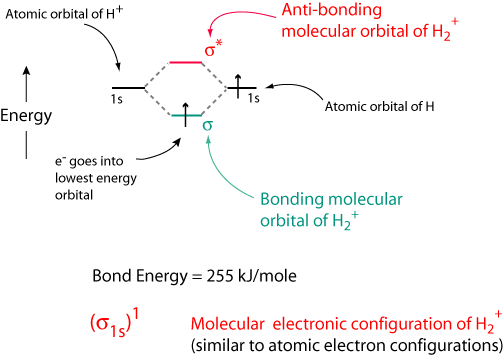
Molecular orbital diagram for he2. On either side of the central ladder are shown the energies of the 1 s orbitals of atoms a and b and the central two rung ladder shows the energies of the bonding and antibonding combinations. Therefore you have two mos one is the lower energy sigma orbital and the other is the higher energy sigma star antibonding orbital. The molecular orbital energy level diagram which is a diagram that shows the relative energies of molecular orbitals for the h 2 molecule is shown in figure 13.
The organic chemistry tutor 266209 views. Fill the molecular orbitals in the energy level diagram beginning with the orbital with the lowest energy. The molecular orbital approach is one explanation for the ceh h bond.
Use an mo diagram to find the bond order and predi. Determine the total number of valence electrons in the he 2 2 ion. Draw the lewis structure of pf 3a how many share.
Combine the two he valence atomic orbitals to produce bonding and antibonding molecular orbitals. Molecular orbital energy level diagram bond order and stability. Fill in the two orbitals following the aufbau principle.
Draw the molecular orbital energy level diagram for the system. Molecular orbital theory bonding antibonding mo bond order homonuclear diatomic molecules duration. Click within the blue boxes to add electrons.
This indicates that koopmans theorem is consistent at all distances and implies that correlation diagrams for neutral molecules should be reasonable approximations to the ionization potential curves. Construct the molecular orbital diagram for he2. When the 1s wave functions of the two ceh atoms are linearly combined we get a sigma s bonding orbital denoted as s 1s in the diagram herethis approach is called linear combination of atomic orbitals lcao.
Molecular orbitals of h 2. 1count the valence electrons that you have for example 3 electrons in he2. Click within the blue boxes to add electrons.
 8 4 Molecular Orbital Theory Chemistry
8 4 Molecular Orbital Theory Chemistry
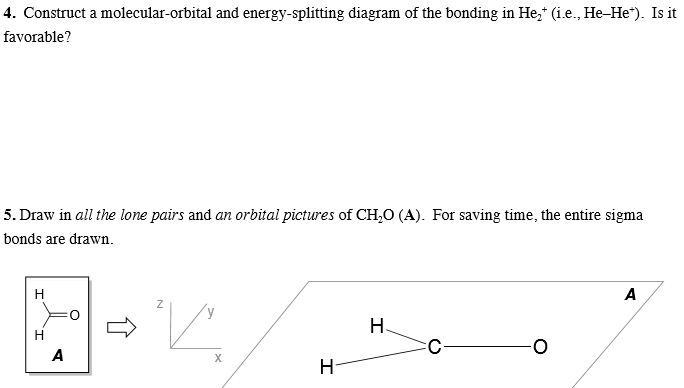 Solved Contruct A Molecular Orbital And Energy Splitting
Solved Contruct A Molecular Orbital And Energy Splitting
Hybridization And Molecular Orbital Mo Theory Pages 1 29
 Audio 03948795249 Bond Order Bonding Electrons Antibonding
Audio 03948795249 Bond Order Bonding Electrons Antibonding
 Use The Molecular Orbital Diagram Shown To Clutch Prep
Use The Molecular Orbital Diagram Shown To Clutch Prep
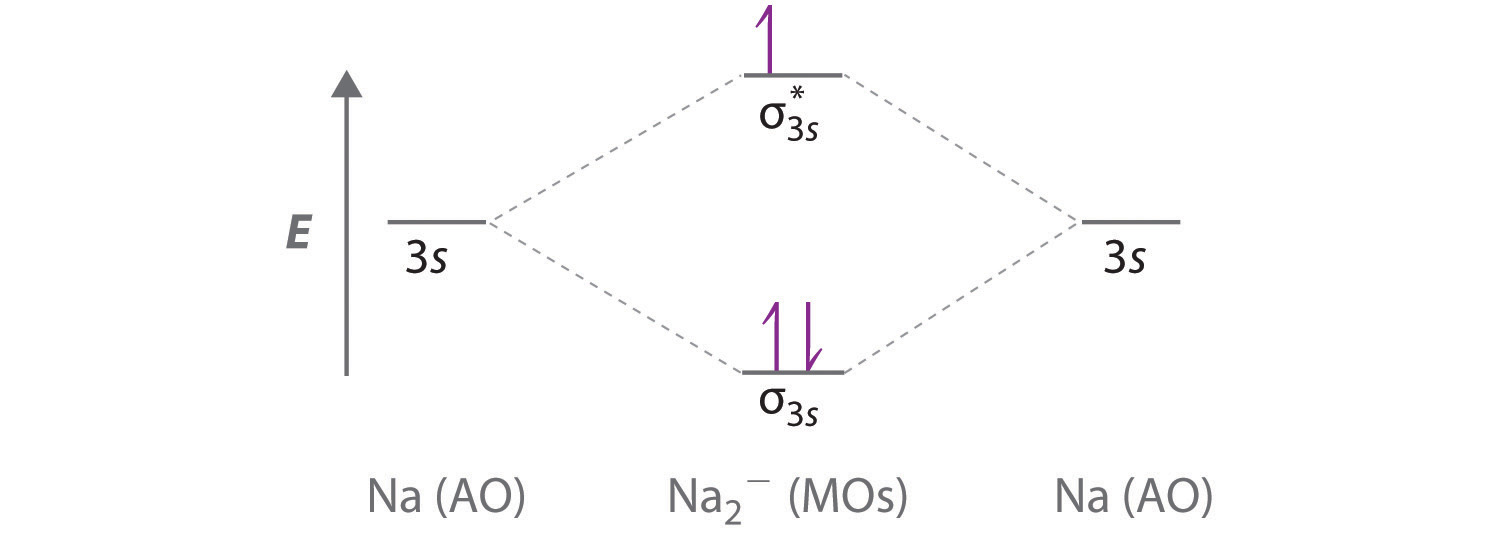 11 5 Molecular Orbital Theory Chemistry Libretexts
11 5 Molecular Orbital Theory Chemistry Libretexts
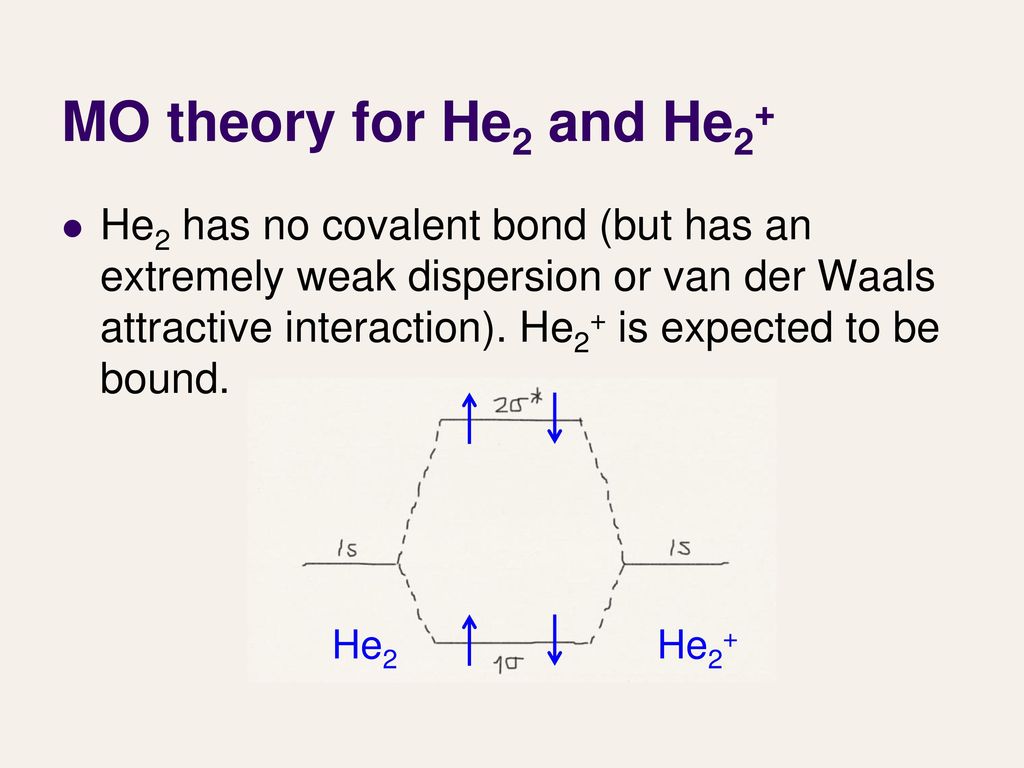 Lecture 27 Molecular Orbital Theory Iii Ppt Download
Lecture 27 Molecular Orbital Theory Iii Ppt Download
 Which Is More Stable He2 Or H2 And Why Quora
Which Is More Stable He2 Or H2 And Why Quora
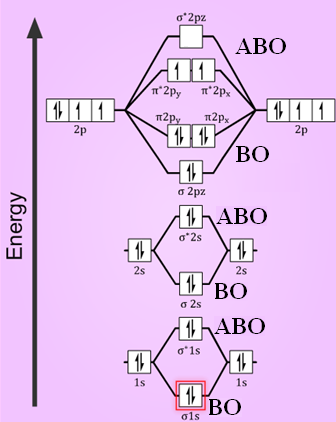 Compare The Stabilities Of O2 O2 O22
Compare The Stabilities Of O2 O2 O22
Orbital Interaction Diagram 1 Plot Atomic Valence Orbital
 Do He2 He2 He2 2 Exist Stable Molecular Orbital Theory
Do He2 He2 He2 2 Exist Stable Molecular Orbital Theory
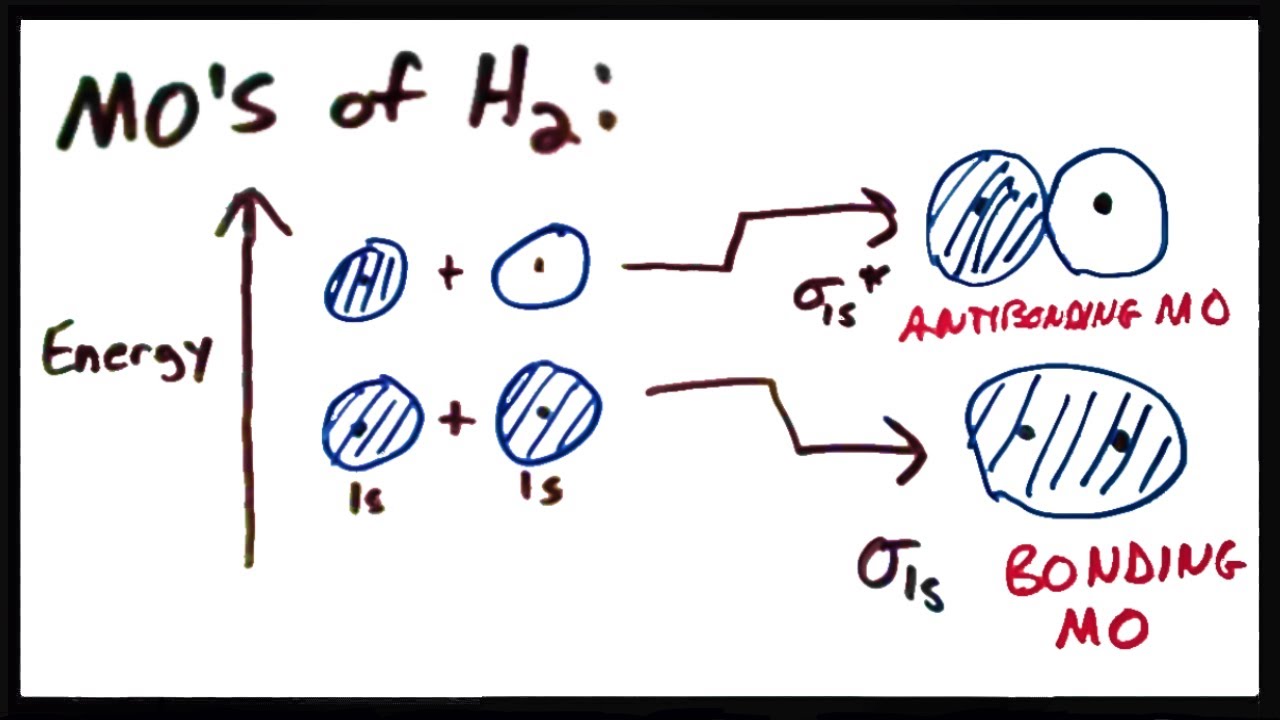
 Molecular Orbital Theory Ii Mo S Of The H2 Molecule
Molecular Orbital Theory Ii Mo S Of The H2 Molecule
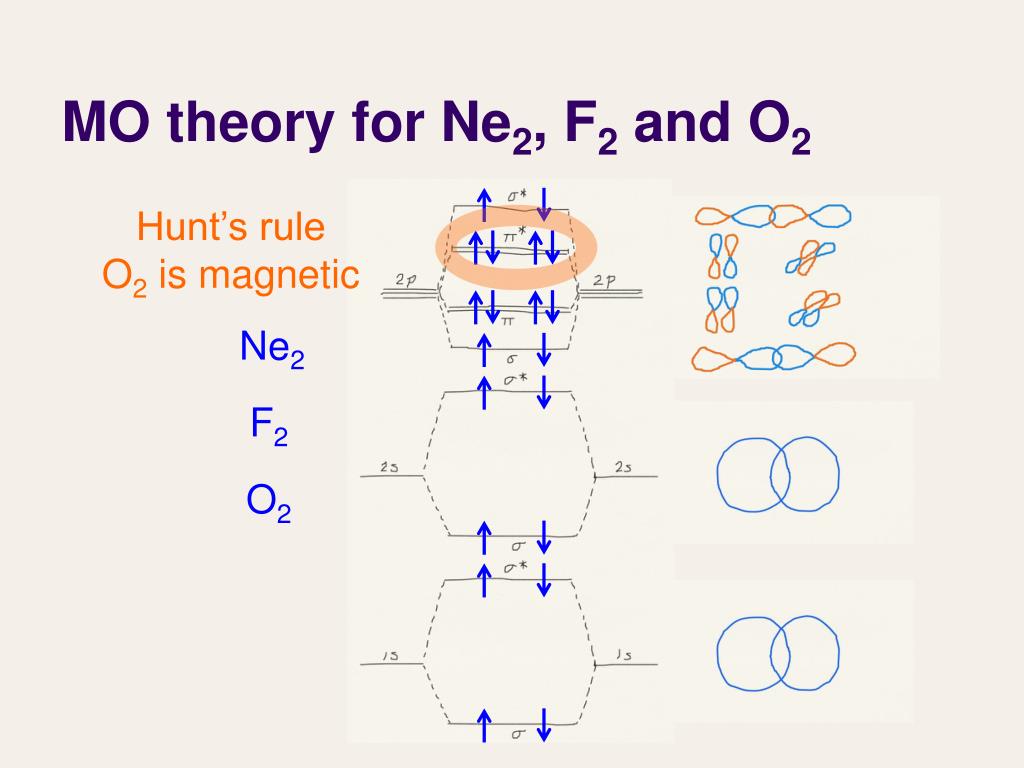 Ppt Lecture 27 Molecular Orbital Theory Iii Powerpoint
Ppt Lecture 27 Molecular Orbital Theory Iii Powerpoint
 Molecular Orbital A Molecule In Which All The Electrons
Molecular Orbital A Molecule In Which All The Electrons
According To Molecular Orbital Theory Which Of The
 By Writing Molecular Orbital Configuration For No Co O2
By Writing Molecular Orbital Configuration For No Co O2
 What Is The Bond Order Of He2 A 0b C 1d 1 E 2
What Is The Bond Order Of He2 A 0b C 1d 1 E 2
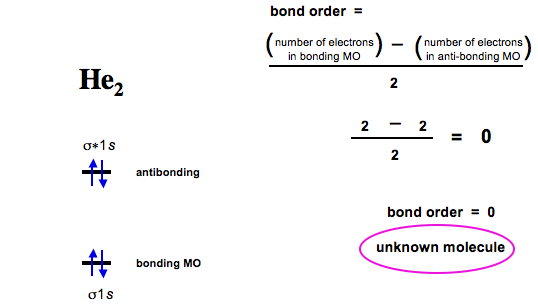 Diatomic Species Mo Theory Chemogenesis
Diatomic Species Mo Theory Chemogenesis
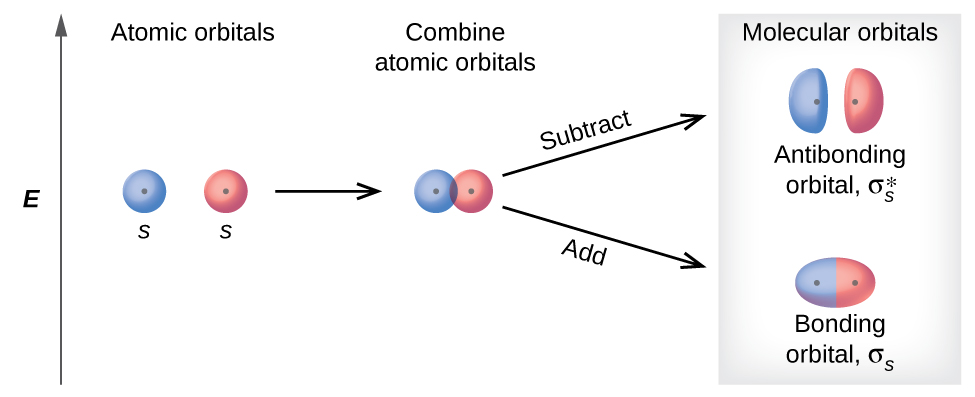 8 4 Molecular Orbital Theory Chemistry
8 4 Molecular Orbital Theory Chemistry
 Chem Test 3 Flashcards Quizlet
Chem Test 3 Flashcards Quizlet
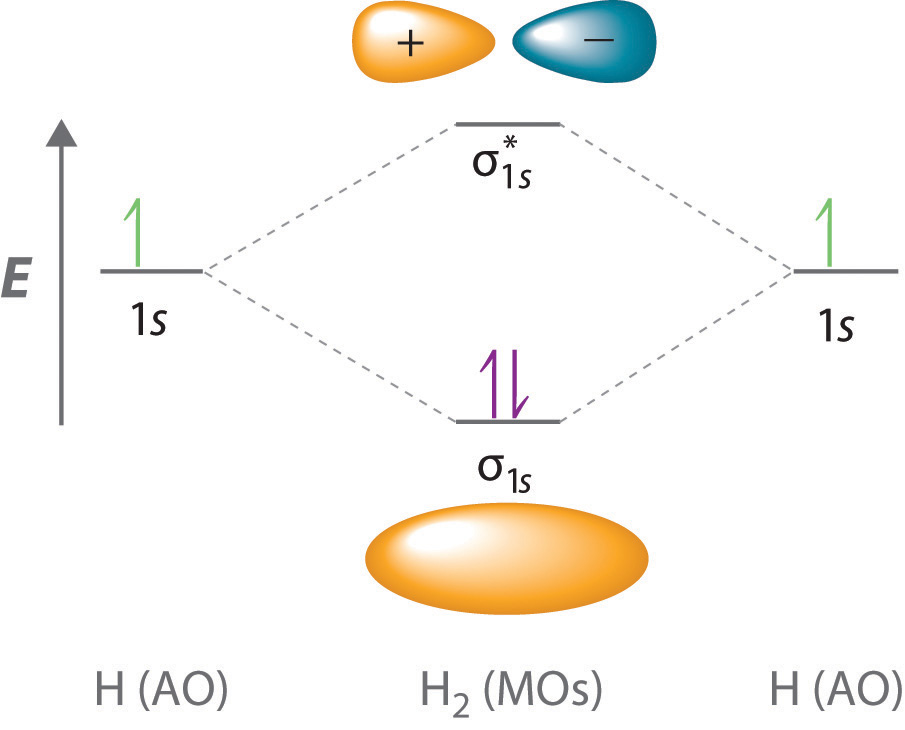 Delocalized Bonding And Molecular Orbitals
Delocalized Bonding And Molecular Orbitals
 By Writing Molecular Orbital Configuration For No Co O2
By Writing Molecular Orbital Configuration For No Co O2
Unit I Molecular Structure And Theories Of Bonding Terms

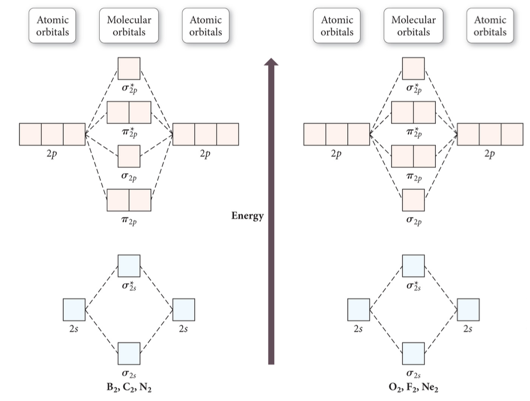
0 Response to "Molecular Orbital Diagram For He2"
Post a Comment