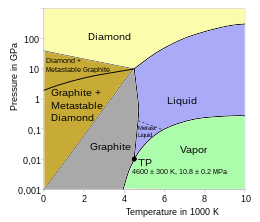
Consider This Phase Diagram For Carbon Which Phases Are Present At The Upper Triple Point
It is worth considering the implications of that difference. Graphite gas diamond liquid which phase is stable at 105 atm and 1000 k.
 Phase Diagrams Chemistry Libretexts
Phase Diagrams Chemistry Libretexts
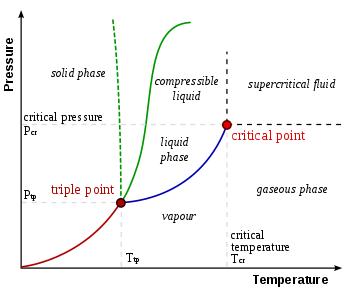
A phase diagram is a graph of the physical state of a substance solid liquid or gas and the temperature and pressure of the substance.

Consider this phase diagram for carbon which phases are present at the upper triple point. Which phases are present at the lower triple point. A phase diagram is a pressure temperature graph th. For most substances the slope of the liquid solid phase equilibrium curve is positive.
This is the phase diagram for a typical pure substance. Gas liquid diamond graphite starting from the lower triple point what actions would produce liquid carbon. There are three solid lines on the phase diagram that are called the phase equilibrium lines.
Graphite 10 gas diamond 1 liquid starting from the lower triple point what actions would produce liquid carbon. Which phases are present at the upper triple point. Consider this phase diagram for carbon.
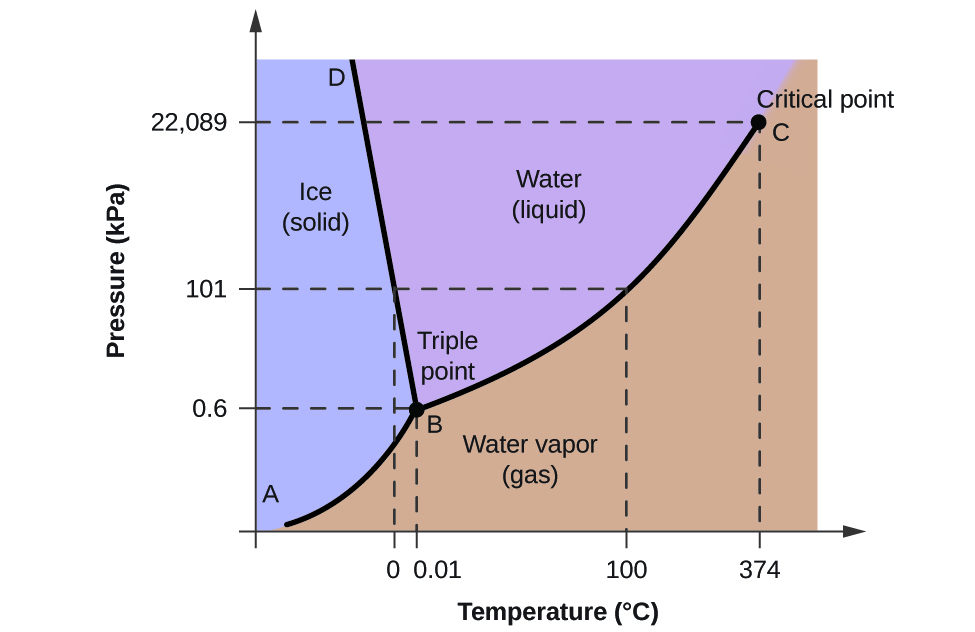
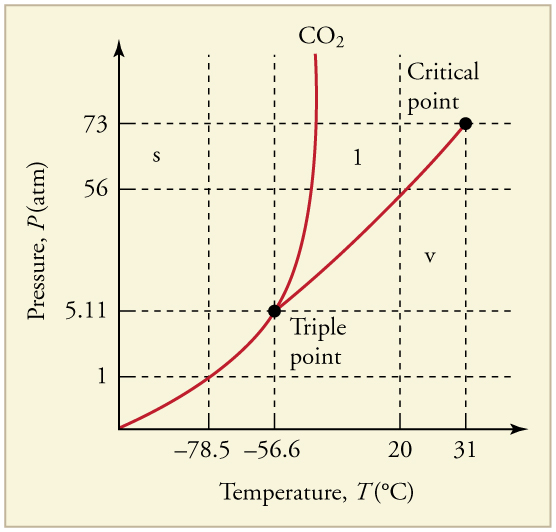
A the upper right region of the phase diagram is the liquid region while the lower right region is the region of gas b melting involves s l. For water the slope is negative. Consider this phase diagram for carbon dioxide in.
Observe that this phase diagram is divided into three regions by the blue boundary lines similar to the way the interactive phase diagram for co2 was divided. As the phase diagram shows the lowest pressure at which liquid exists is at the triple point pressure 43atm. Phase diagrams a phase diagram lets you work out exactly what phases are present at any given temperature and pressure.
Consider the following phase diagram and identify. Compare this general form of this phase diagram with the interactive phase diagram for co2 and classify points a through g on the phase diagram according to the number of phases present at each point. Consider this phase diagram for carbon.
These lines show where two phases are in equilibrium. 100atm is far below 43 atm. In the cases well be looking at on this page the phases will simply be the solid liquid or vapour gas states of a pure substance.
We are finally in a position to define the triple point it is just the point at which the two phase equilibrium lines intersect. All substances except helium have triple points. Diamond graphite gas liquid which phase is stable at 105 atm and 1000 k.
 Consider This Phase Diagram For Carbon Wh Clutch Prep
Consider This Phase Diagram For Carbon Wh Clutch Prep
Triple Point Phase Transition For Carbon Dioxide
 Consider This Phase Diagram For Carbon Wh Clutch Prep
Consider This Phase Diagram For Carbon Wh Clutch Prep
13 5 Phase Changes Texas Gateway
 Consider This Phase Diagram For Carbon Wh Clutch Prep
Consider This Phase Diagram For Carbon Wh Clutch Prep
 Phase Diagrams Of Water Co2 Explained Chemistry Melting Boiling Critical Point
Phase Diagrams Of Water Co2 Explained Chemistry Melting Boiling Critical Point
 Phase Diagrams Video States Of Matter Khan Academy
Phase Diagrams Video States Of Matter Khan Academy
 Consider This Phase Diagram For Carbon Which Phases Are Present At The Lower Triple Point A Diamondb Graphitec Gasd Liquid
Consider This Phase Diagram For Carbon Which Phases Are Present At The Lower Triple Point A Diamondb Graphitec Gasd Liquid
 Solved Consider This Phase Diagram For Carbon Which Phas
Solved Consider This Phase Diagram For Carbon Which Phas
Teach Yourself Phase Diagrams And Phase Transformations
 Solubility Of Co2 In 1 N Butylthiolanium Tf2n Toluene
Solubility Of Co2 In 1 N Butylthiolanium Tf2n Toluene
 2 Carbon Dioxide Co2 Pressure Temperature Phase Diagram
2 Carbon Dioxide Co2 Pressure Temperature Phase Diagram
 Consider This Phase Diagram For Carbon Wh Clutch Prep
Consider This Phase Diagram For Carbon Wh Clutch Prep
 Stability Of Dense Liquid Carbon Dioxide Pnas
Stability Of Dense Liquid Carbon Dioxide Pnas
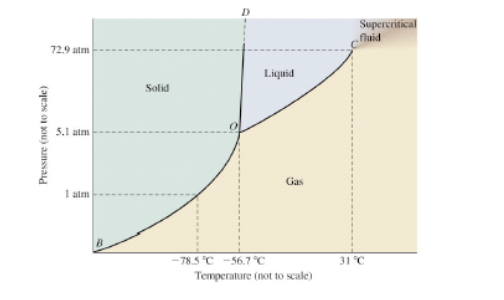 Consider This Phase Diagram For Carbon Wh Clutch Prep
Consider This Phase Diagram For Carbon Wh Clutch Prep
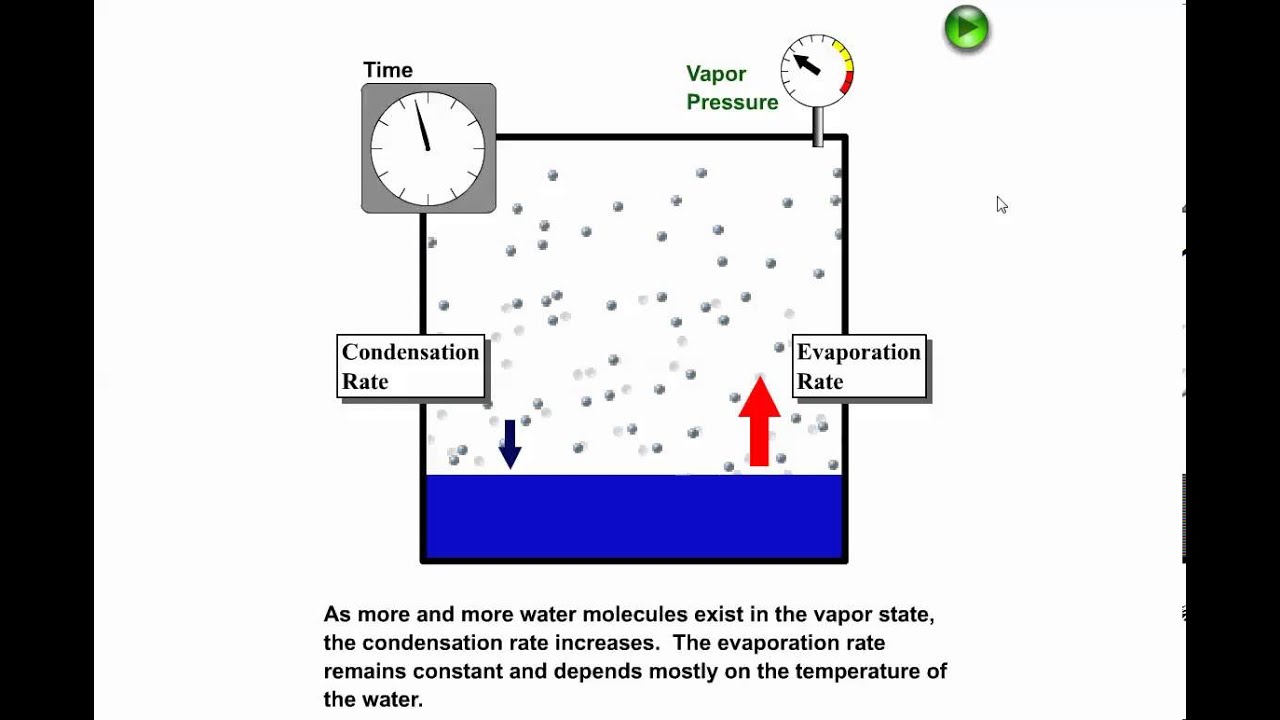 Phase Changes Boundless Chemistry
Phase Changes Boundless Chemistry
 Solved Consider This Phase Diagram For Carbon Which Phas
Solved Consider This Phase Diagram For Carbon Which Phas
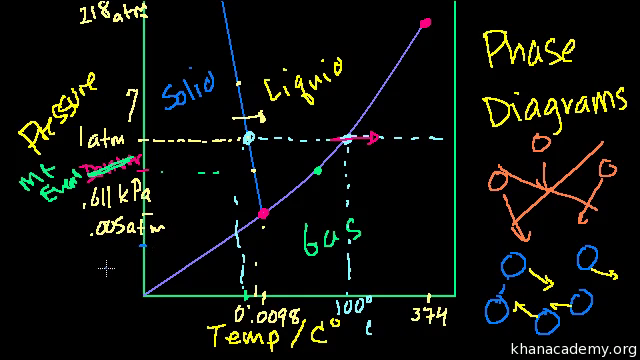 Phase Diagrams Video States Of Matter Khan Academy
Phase Diagrams Video States Of Matter Khan Academy
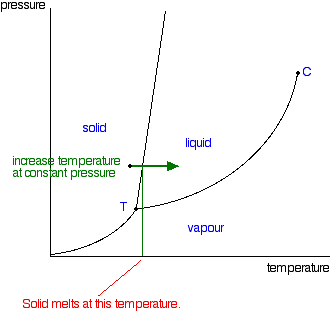 Phase Diagrams Of Pure Substances
Phase Diagrams Of Pure Substances
Iron Carbon Phase Diagram A Review See Callister Chapter 9

0 Response to "Consider This Phase Diagram For Carbon Which Phases Are Present At The Upper Triple Point"
Post a Comment