What Is An Orbital Diagram
The aufbau principle the pauli exclusion principle and hunds rule. The number of valence electrons impacts on their chemical properties and the specific ordering and properties of the orbitals are important in physics so many students have to get to grips with the basics.
 Molecular Orbital Diagram Corresponding To An Octahedral Mx
Molecular Orbital Diagram Corresponding To An Octahedral Mx
Three rules are useful in forming orbital diagramsaccording to the auf bau principle each electron occupies the lowest energy orbital.

What is an orbital diagram. Each orbital can hold 2 electrons and each arrow. An orbital may also be called an atomic orbital or electron orbital. Two atomic orbitals in phase create a larger electron density which leads to the σ orbital.
Many times it is necessary to see all the quantum numbers in an electron configuration this the purpose of the orbital diagram. An orbital filling diagram is the more visual way to represent the arrangement of all the electrons in a particular atom. In an orbital filling diagram the individual orbitals are shown as circles or squares and orbitals within a sublevel are drawn next to each other horizontally.
Electron orbital diagrams and written configurations tell you which orbitals are filled and which are partially filled for any atom. The superposition of the two 1s atomic orbitals leads to the formation of the σ and σ molecular orbitals. For the diagram you start with the 1 s orbital and then 2s 2p and so on.
Orbital diagrams must follow 3 rules. Each orbital is shown as a single square and orbitals within the same sub level are drawn directly next to each other. Orbital diagrams are pictorial descriptions of the electrons in an atom.
Each orbital in an atom is characterized by a unique set of values of the three quantum numbers n ℓ and m which respectively correspond to the electrons energy angular momentum and an angular momentum vector component the magnetic quantum number. Orbital diagrams are a visual way to show where the electrons are located within an atom. Although most people think of an orbit in terms of a circle the probability density regions that may contain an electron may be spherical dumbbell shaped or more complicated three dimensional forms.
In addition to listing the principle quantum number n and the subshell ell the orbital diagram shows all the different orientations and the spin of every electron. An orbital diagram provides a visual representation of the way in which an atoms electrons are distributed into various orbitals. If the two 1s orbitals are not in phase a node between them causes a jump in energy the σ orbital.
The pauli exclusion principle says that only two electrons can fit into an single orbital. An orbital is a region of space that an electron can exist in.
 An Example Of A Frontier Molecular Orbital Diagram Diagram
An Example Of A Frontier Molecular Orbital Diagram Diagram
 What Is The Orbital Diagram For Calcium Study Com
What Is The Orbital Diagram For Calcium Study Com
 Orbital Diagram For Ca2 Wiring Diagram
Orbital Diagram For Ca2 Wiring Diagram
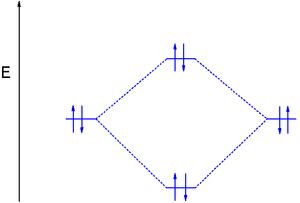 Molecular Orbital Diagram Wikipedia
Molecular Orbital Diagram Wikipedia
 Qualitative Molecular Orbital Diagram Of Hf The 2s
Qualitative Molecular Orbital Diagram Of Hf The 2s
 How Do You Write The Orbital Diagram For Phosphate Socratic
How Do You Write The Orbital Diagram For Phosphate Socratic
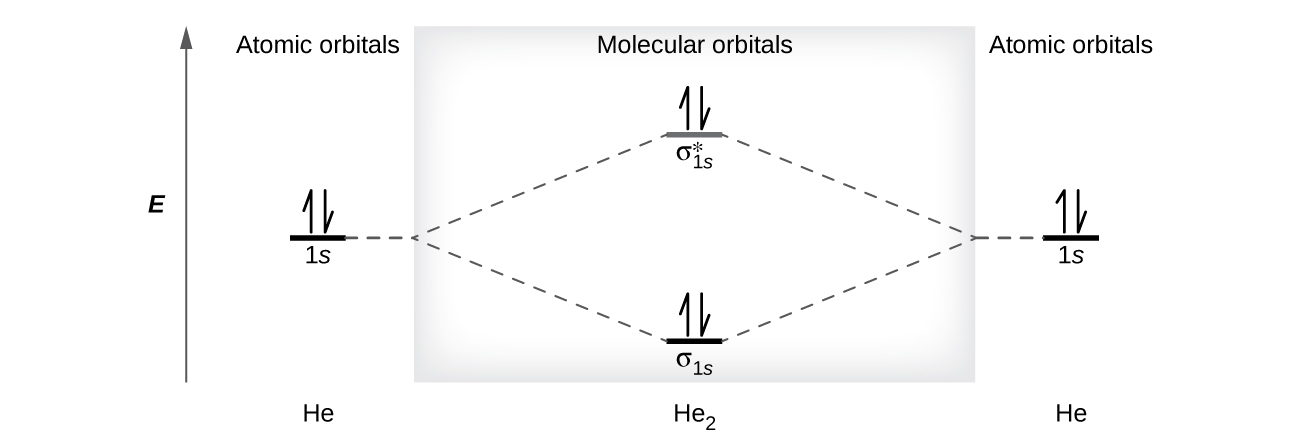 8 4 Molecular Orbital Theory Chemistry
8 4 Molecular Orbital Theory Chemistry
 File Palladium Ligand Molecular Orbital Diagram Png
File Palladium Ligand Molecular Orbital Diagram Png
 Orbital Filling Diagrams The Cavalcade O Chemistry
Orbital Filling Diagrams The Cavalcade O Chemistry
 What Is The Orbital Diagram For Vanadium Quora
What Is The Orbital Diagram For Vanadium Quora
 Orbital Diagrams Electron Configurations For Atoms And Ions
Orbital Diagrams Electron Configurations For Atoms And Ions
 Theoretical Chemistry Energetic Placement Of Atomic
Theoretical Chemistry Energetic Placement Of Atomic
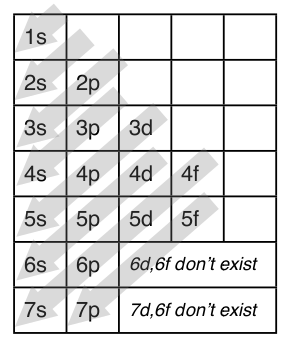 1 4 Electron Configuration And Orbital Diagrams Chemistry
1 4 Electron Configuration And Orbital Diagrams Chemistry
 High School Chemistry Orbital Configurations Wikibooks
High School Chemistry Orbital Configurations Wikibooks
 Electron Orbital Diagram Of Vanadium Chemistry Stack Exchange
Electron Orbital Diagram Of Vanadium Chemistry Stack Exchange
 Molecular Orbital Diagram Of Cof 6 3 Complex With Six P
Molecular Orbital Diagram Of Cof 6 3 Complex With Six P
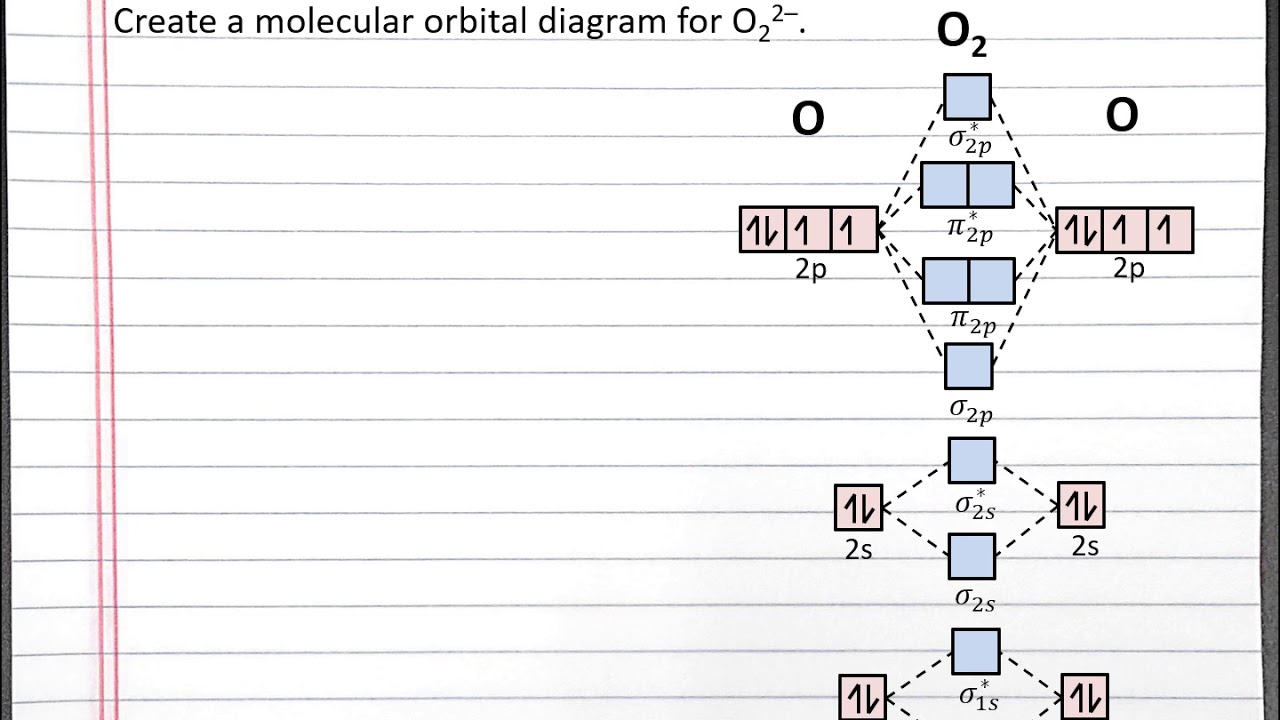 Chem 101 Creating A Molecular Orbital Diagram For A Diatomic Ion In The Second Row With Aleks
Chem 101 Creating A Molecular Orbital Diagram For A Diatomic Ion In The Second Row With Aleks
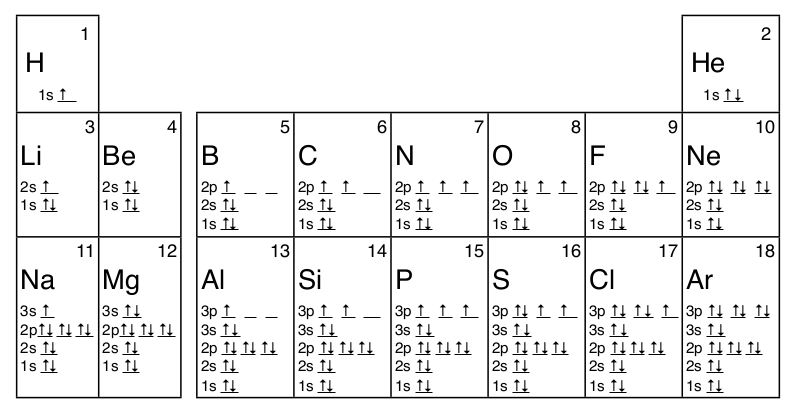 1 4 Electron Configuration And Orbital Diagrams Chemistry
1 4 Electron Configuration And Orbital Diagrams Chemistry
 Mathematics Origins Of Molecular Orbital Diagrams
Mathematics Origins Of Molecular Orbital Diagrams
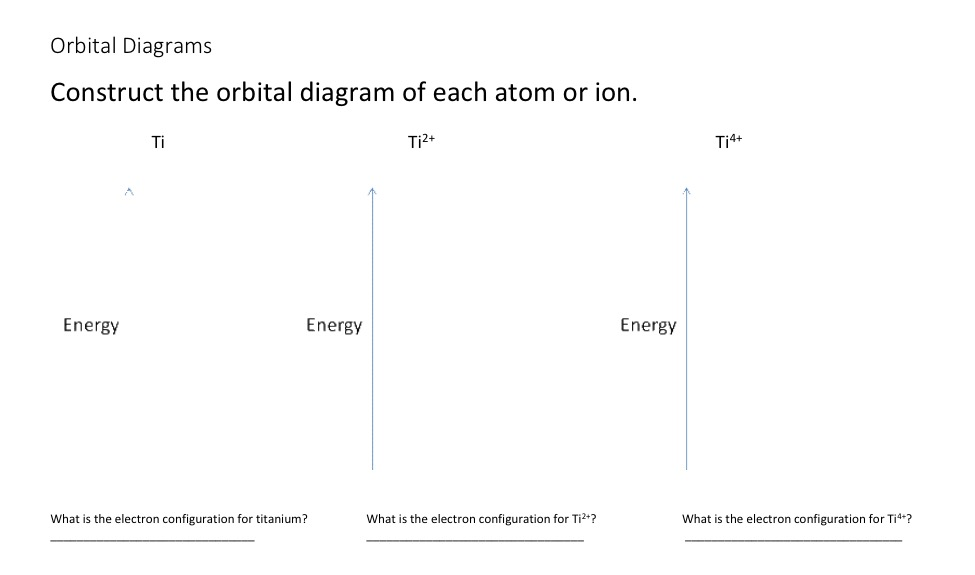 Solved Construct The Orbital Diagram Of Each Atom Or Ion
Solved Construct The Orbital Diagram Of Each Atom Or Ion
 Orbital Diagram Cl Group Electrical Schemes
Orbital Diagram Cl Group Electrical Schemes
 Draw The Molecular Orbital Diagram Of O2 Or N2 Brainly In
Draw The Molecular Orbital Diagram Of O2 Or N2 Brainly In
 Quiz Worksheet Practice Drawing Electron Orbital
Quiz Worksheet Practice Drawing Electron Orbital

0 Response to "What Is An Orbital Diagram"
Post a Comment