Lewis Dot Diagram For Co2
In the co 2 lewis structure carbon is the least electronegative element. This is the lewis dot structure for carbon dioxide.
 Dot Diagram Of Co2 Wiring Diagrams Folder
Dot Diagram Of Co2 Wiring Diagrams Folder
Each oxygen in this has two lone pairs and has a double connection with the carbon.

Lewis dot diagram for co2. There is an easy way and a formal way to draw the lewis structure of co 2 carbon dioxide. Lewis structures also known as lewis dot structures or electron dot structures are diagrams that represent the valence electrons of atoms within a molecule. This is the lewis dot structure for carbon dioxide.
This info can then be used to determine the lewis dot structure. Both oxygens have 6e and carbon has 4. This gives both.
The lewis dot structure is a graphical representation of how electrons are distributed around the atoms which comprise a molecule. Therefore it is put in the center of the dot structure. Both oxygens have 6e and carbon has 4.
For the co 2 lewis structure there are a total of 16 valence electrons available. These lewis symbols and lewis structures help visualize the valence electrons of atoms and molecules whether they exist as lone pairs or within bonds. If you are new in the field of molecular geometry and lewis structure firstly you should understand the meaning of lewis structure for the better understanding of the molecules shape.
In the earths atomsphere it is considered a greenhouse gas. Lewis structure is nothing but the electron structure made by the dots. The reason for drawingcreating a lewis dot structure is that it helps one predict the kinds of bonds as well as a number of bonds that can be formed around an atom.
Each oxygen in this has two lone pairs and has a double connection with the carbon. Therefore it is nonpolar and relatively unreactive. In the formal way we find how many electrons we have step 1 how many each atom needs step 2 how many of those are bonding step 3 4 and how many are lone pairs step 5.
The lewis dot structure of co2 gives it some unique properties. Co 2 is a clear heavier than air gas. Since there are no lone pairs on the atom it is a linear structure which makes the charges cancel it.
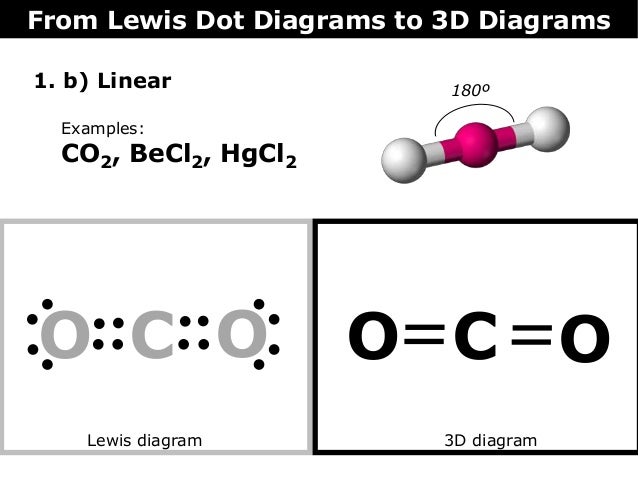 Co2 Dot Diagram Catalogue Of Schemas
Co2 Dot Diagram Catalogue Of Schemas
 Lewis Dot Structure Of Co2 3 Chemistry Chemical Bonding
Lewis Dot Structure Of Co2 3 Chemistry Chemical Bonding
 The Lewis Dot Structure For Co2 Makethebrainhappy
The Lewis Dot Structure For Co2 Makethebrainhappy
Geometry Of Molecules Chemistry Libretexts
 Co2 Dot Diagram Catalogue Of Schemas
Co2 Dot Diagram Catalogue Of Schemas
 Dot Diagram Of Co2 Review From The End Of Fall Semester
Dot Diagram Of Co2 Review From The End Of Fall Semester
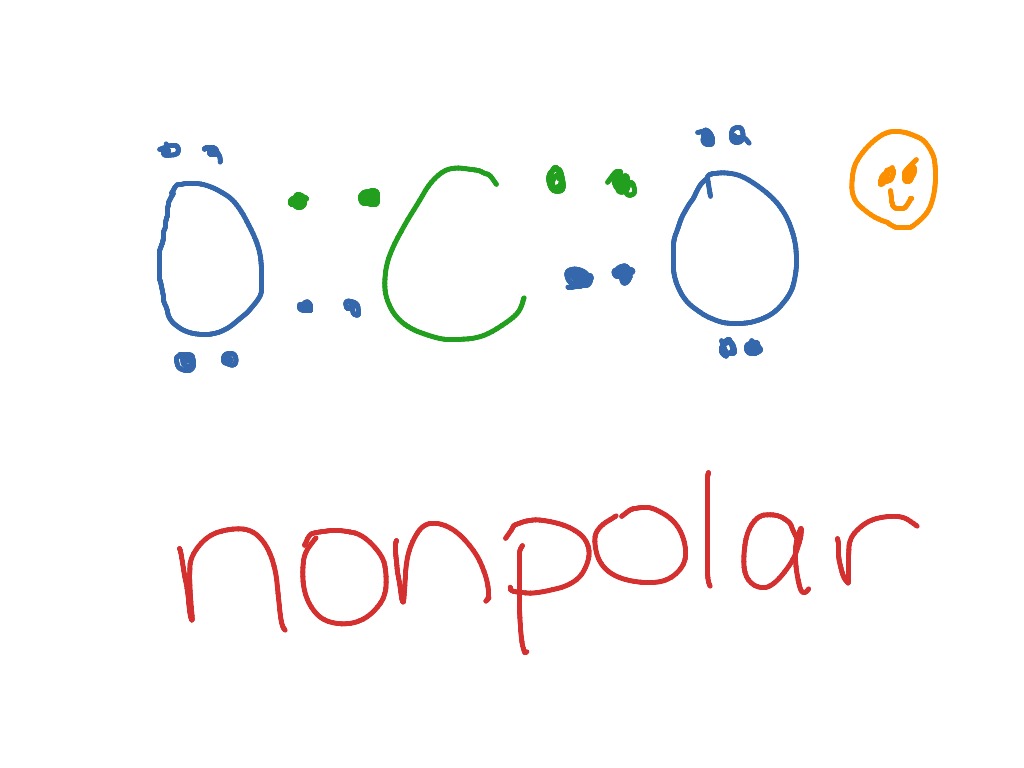 Dot Diagram Of Co2 Wiring Diagram
Dot Diagram Of Co2 Wiring Diagram
 Co2 Dot Diagram Wiring Diagram Schematics
Co2 Dot Diagram Wiring Diagram Schematics
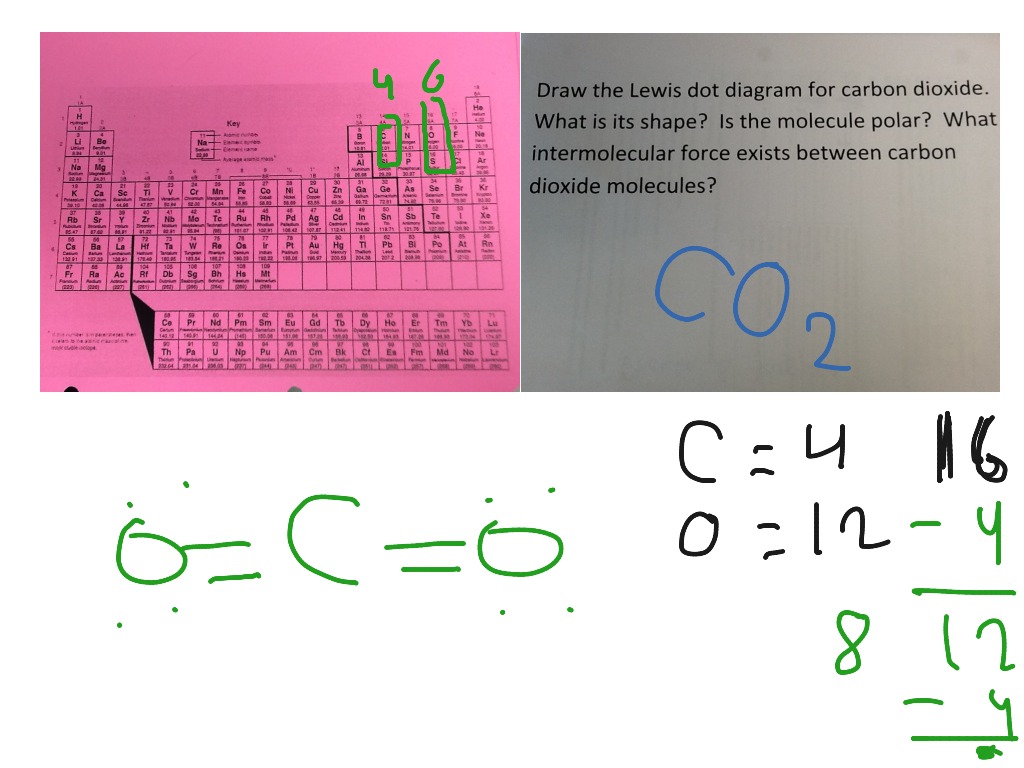 Lewis Dot Diagram Of Co2 Science Showme
Lewis Dot Diagram Of Co2 Science Showme
 Dot Diagram Of Co2 Wiring Diagram Schematics
Dot Diagram Of Co2 Wiring Diagram Schematics
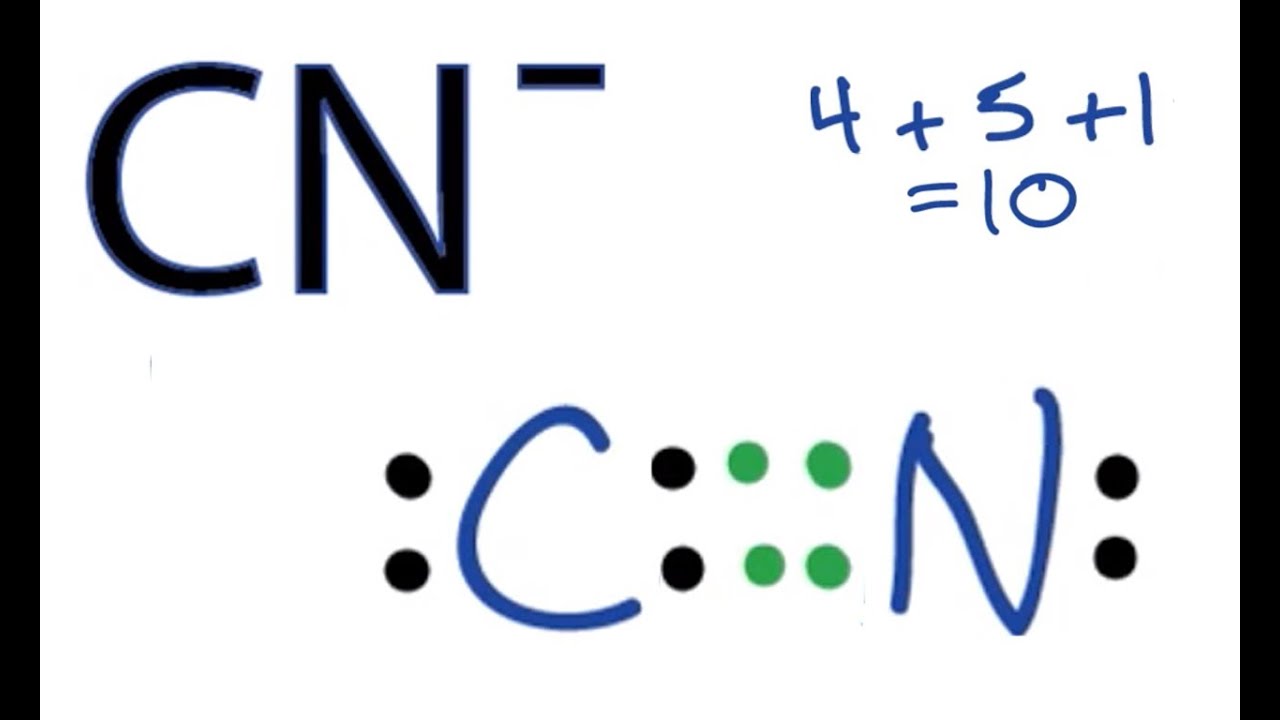 The Lewis Dot Structure For Co2 Makethebrainhappy
The Lewis Dot Structure For Co2 Makethebrainhappy
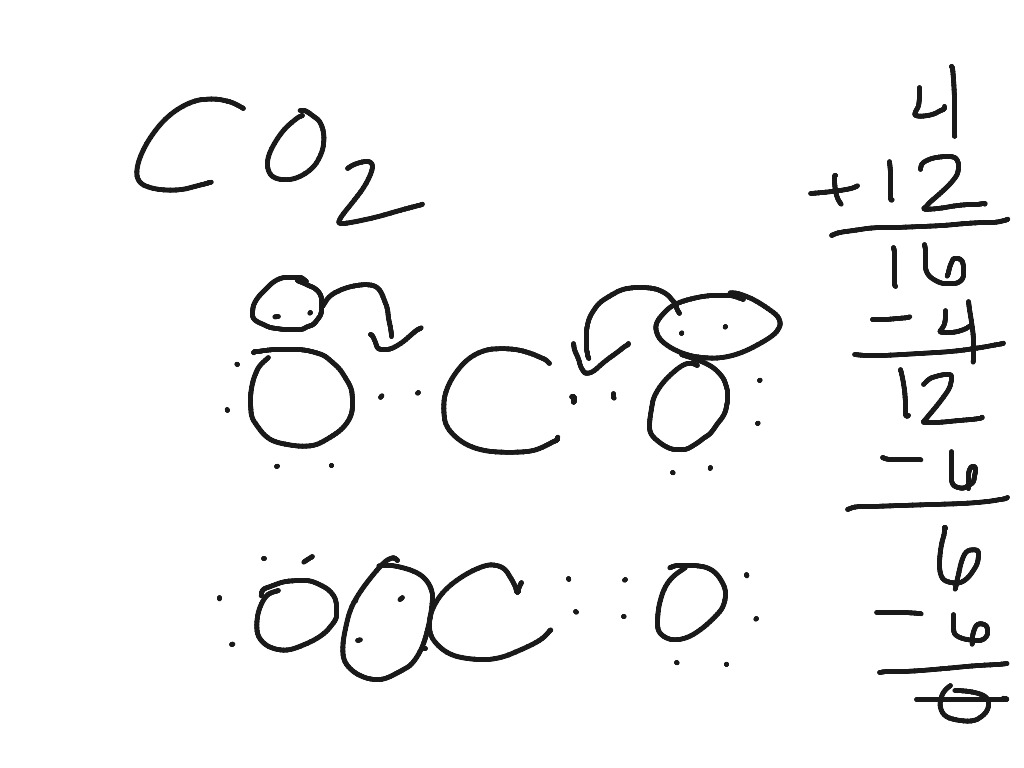 Lewis Dot Structure Co2 Science Chemistry Chemical Bonds
Lewis Dot Structure Co2 Science Chemistry Chemical Bonds
 Co2 Dot Diagram Catalogue Of Schemas
Co2 Dot Diagram Catalogue Of Schemas
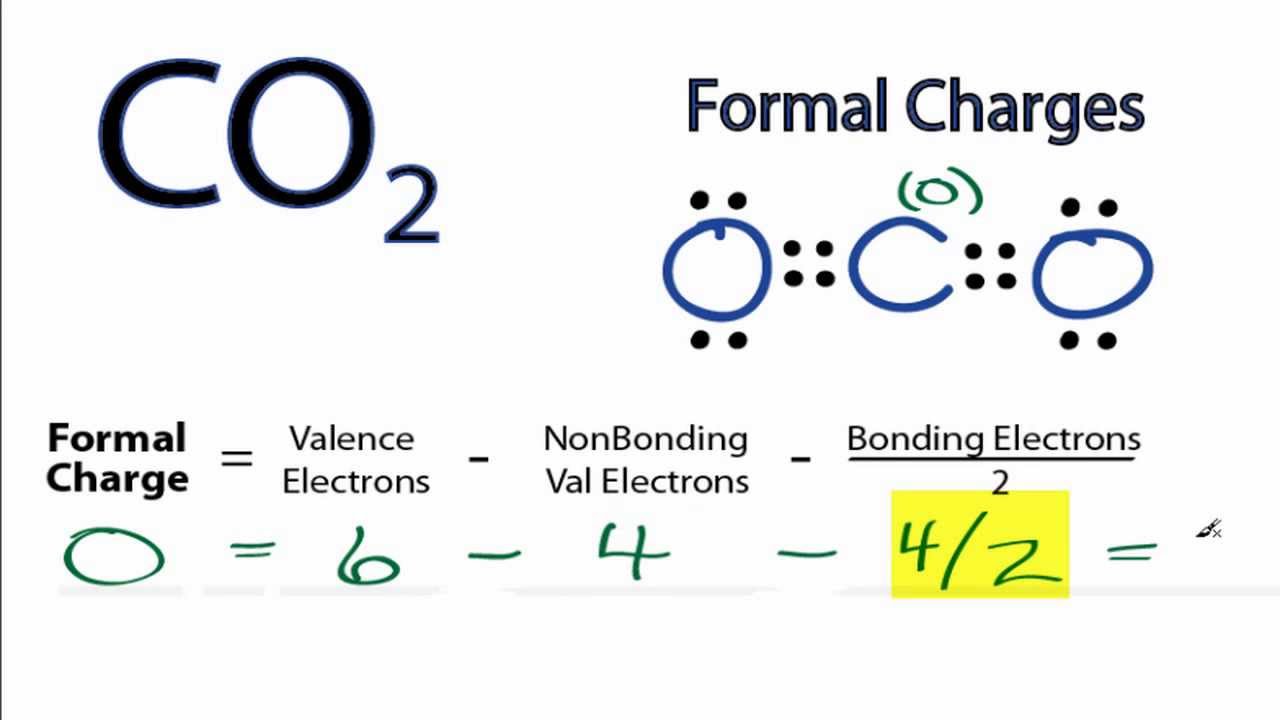 Co2 Dot Diagram Catalogue Of Schemas
Co2 Dot Diagram Catalogue Of Schemas
 The Lewis Dot Structure For Co2 Makethebrainhappy
The Lewis Dot Structure For Co2 Makethebrainhappy
 The Lewis Dot Structure For Co2 Makethebrainhappy
The Lewis Dot Structure For Co2 Makethebrainhappy
 Co2 Lewis Structure How To Draw The Dot Structure For
Co2 Lewis Structure How To Draw The Dot Structure For
 Co2 Lewis Structure How To Draw The Dot Structure For
Co2 Lewis Structure How To Draw The Dot Structure For
 Co2 Molecular Geometry And Lewis Structure
Co2 Molecular Geometry And Lewis Structure
 Electron Dot And Structual Diagrams For Covalent Compounds
Electron Dot And Structual Diagrams For Covalent Compounds
Co2 Dot Diagram Catalogue Of Schemas
 Lewis Diagram Co Catalogue Of Schemas
Lewis Diagram Co Catalogue Of Schemas
How Can I Draw A Lewis Dot Diagram For Carbon Dioxide
 Lewis Dot Symbols And Lewis Structures Boundless Chemistry
Lewis Dot Symbols And Lewis Structures Boundless Chemistry
 Co2 Carbon Dioxide Molecule One Carbon Atom With Two
Co2 Carbon Dioxide Molecule One Carbon Atom With Two

0 Response to "Lewis Dot Diagram For Co2"
Post a Comment